
|
Software + Services + Expertise |

|

KnarrTek provides affordable CFR 21 Part 11 compliant software that can be used to track the manufacture, repackaging, and distribution of pharmaceutical products.
This software supports DSCSA Serialization tracking, including tracking serial numbers, printing barcode labels, and tracking which ingredients went into each batch of products.
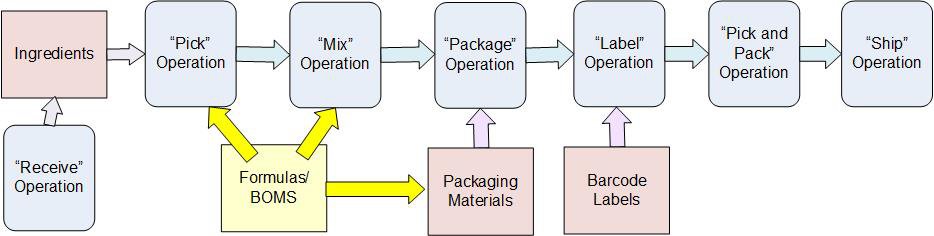
The KnarrTek software captures complete end-to-end traceability data to support tracking which ingredients went into which serialize products as well as the equipment and people involved. It also tracks how the products were packed and who they were shipped to.
The KnarrTek software can track the receipt and put-away of ingredients by lot and serial number as well as tracking expiration dates and ensuring that quality control inspection is performed before the ingredients are used.
The KnarrTek software can then track the picking of products for each batch, warning material handlers if the wrong materials are picked for the manufacture of a batch of materials.
The KnarrTek software tracks serial numbers for products and can print out rolls of serial number labels for use in automated applicators or feed the serial numbers to automated labeling equipment in a process line.
The KnarrTek software can then track which boxes each serial numbered product was placed into and which pallets the boxes were placed on. It can also record the picking of products for customer orders into mixed product boxes and pallets.
The KnarrTek software; is GS1 compliant. Through the use of the MilramX it can generate ASN (Advanced Shipment Notice) data for transmission to customers by third party EDI applications. It can also receive ASN data from EDI applications for use in efficient receiving of materials.
For more information about how KnarrTek can help you with your specific pharmaceutical manufacturing or materials traceability issues, please contact KnarrTek by clicking on the [Contact Us] button below:
| About KnarrTek | Technology |

|
Services | Cost Savings |
| Copyright © KnarrTek Inc. 2024 |
| Technology |

|
Services |
| About KnarrTek | Copyright © KnarrTek Inc. 2024 | Cost Savings |